Back to your search result
Description
Note: Pharmaceutical Analytical Impurities are released using a process developed by USP's subject matter experts. The release process is based on internal policies, standard operating procedures, and requirements as defined by USP's Quality Management System. USP is an ISO 9001:2015 certified facility. PAI products are different from official USP Reference Standards. PAI products are not required for compendial compliance.
Structure formula
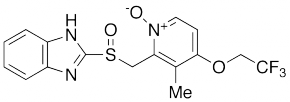
Contents
Miscellaneous
Certificate of Analysis (specimen)
Reference Material CoA specimen: for the current lot, please contact your customer service representative at info@labmix24.com
Product data sheet
View all available product details e.g. description, analytes/parameters, CAS Number, concentrations/values, sales unit/product format, method, source, transport information
US Pharmacopeia Standards set a global benchmark, enabling efficient, reliable, and cost-effective development and quality control of pharmaceuticals.
Official USP Reference Standards are highly characterized physical specimens intended for quality control use when conducting assays and tests for medicines as described in the USP-NF. When you conduct tests and assays required by the USP-NF using the USP reference standards specified, the results can be considered conclusive.
As a USP-Authorized Distributor, we offer the following products and services:
- USP Reference Standards
- USP Pharmaceutical Analytical Impurities
- USP Training and educational programs